EMS was dispatched to a 48-year-old female having a seizure.
As they walked into the residence the patient was laying on the couch, not responding to commands. Her carotid pulse was palpable but the radial was absent. She was noted to be cyanotic and breath sounds were clear.
Side stream ETCO2 was placed under a non-rebreather (12 mm Hg) and rapid crystalloid infusion was initiated via an 18G IV.
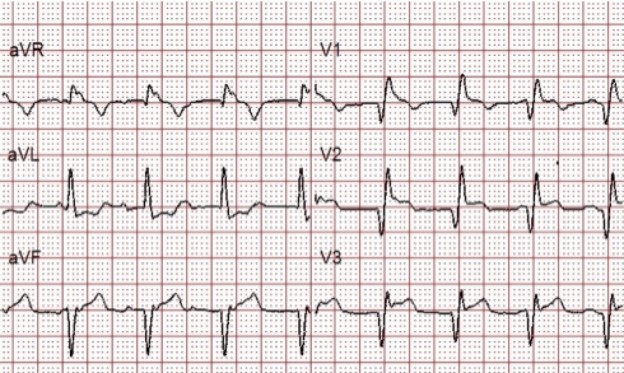
A 12 Lead was obtained as another first responder went to retrieve the stretcher:
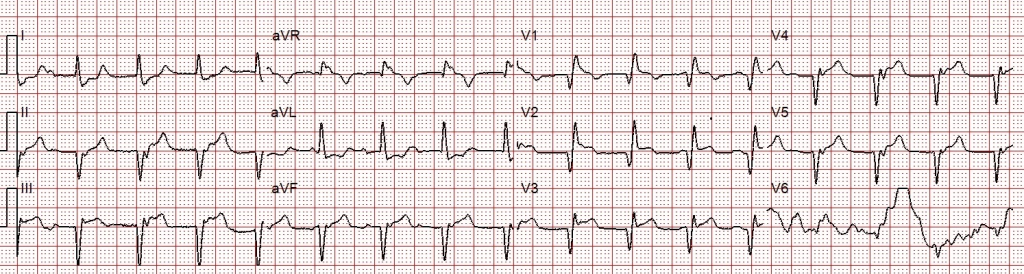
The QRS is wide (160ms) with RBBB morphology (QR in V1). Q-waves and ST-segment elevation are noted in the inferior and anterior leads.
Is this patient having a STEMI?
While the patient was being treated and extricated, a history was obtained from the husband. She had previously been on anticoagulation for a deep venous thrombosis that occurred secondary to hormone therapy. Combining the patient’s history with clinical findings, acute pulmonary embolism was suspected.
Clinical Evaluation of Suspected Pulmonary Embolism
The mortality rate for pulmonary embolism is 10% when diagnosed, but less than 50% are diagnosed at presentation. While there are many signs and symptoms of acute PE, 30% will have no perception of pain, but 90% will admit to a sensation of dyspnea.
Stephen Smith, M.D. presented a similar case recently and made these useful observations:
- Hypoxia with clear lungs is pulmonary embolism until proven otherwise
- STEMI only causes hypoxia by causing pulmonary edema
- Massive pulmonary embolism can result in a STEMI ECG, identical to ACS STEMI
- Low end tidal CO2 is typical of massive PE. High end tidal CO2 is typical of severe pulmonary edema.
Using the ECG to Diagnose a Pulmonary Embolism
The ECG cannot “make the diagnosis” of PE, but can certainly be used to bolster the diagnosis and prompt further evaluation. Changes in the ST/T waves are normally seen in massive PE, and when present can help identify patients that are likely to deteriorate if immediate intervention is not performed. Had an appropriate history not been obtained in the case above, the ST-segment elevation could have been considered a coronary occlusion.
While many are familiar with the ECG finding known as “S1Q3T3” (S-wave in lead 1, Q-wave in lead III, and inverted T-wave in lead III), in a study comparing the incidence of various ECG findings in PE, Chan TC (2001) found that this finding is present only 11-50% of the time.
Sinus tachycardia is the most common ECG finding and is present 8-69% of the time, T-wave inversions in leads III and aVF are present 17-35% of the time, and T-wave inversions in leads V1 and V2 are present 27-68% of the time. Any time you see inferior or anterior T-wave inversions you should consider acute pulmonary embolism in your differential diagnosis.
This ECG is not a representative case of these changes but for a detailed explanation of ECG changes associated with PE you can review Dr. Ken Grauer’s ECG Blog #119.
Ischemia in Acute Pulmonary Embolism
Under normal physiological conditions, the right ventricle has very little resistance relative to the left ventricle. During acute, massive PE, there is increased right ventricular afterload that causes right ventricular dilation and an increasing workload that leads to severe demand ischemia of the right ventricle, which is already prone to ischemia because of the non-dominant blood supply.
It can be extremely difficult at times to distinguish between acute coronary occlusion and PE, which is why prehospital history can be invaluable. Flight Surgeon Dr. Samuel M Galvagno said:
“Indeed, in the management of PE, the prehospital provider’s history may provide the most important clue to the underlying disease process for patients who deteriorate before interrogation by emergency department staff”.
ETCO2 in Acute Pulmonary Embolism
When a blood clot occludes a pulmonary artery the lack of blood flow to the alveoli causes an increase in dead space ventilation. Areas of the lungs that once participated in gas exchange can no longer do so. Clinically, the SpO2 will remain low or not reflective of the amount of oxygenation. As dead space increases, ETCO2 decreases.
Case Conclusion
Immediately before transport the patient became extremely bradycardic and with agonal respirations. She was promptly intubated and given 1 mg of Atropine. The heart rate significantly improved but she remained in profound shock and cyanotic despite maximal oxygen therapy.
During transport she lost a pulse and had an asystolic arrest. Transport time to the ER was approximately 1.5-2 minutes, so chest compressions were started and the receiving facility was notified of the probable massive pulmonary embolism and cardiac arrest.
The receiving physician agreed that PE was the likely diagnosis. It just so happened that an interventionalist was in the ER and was pulled over to assist in the resuscitation. ROSC was achieved and an echo was performed showing acute right heart strain.
The patient was transported to the operating theater. Direct intra-arterial tPA was administered and clot extraction was attempted. The patient rearrested and further attempts at resuscitation was unsuccessful.
References
1.) Brady, William, and Jonathon Dean Truwit. Critical Decisions In Emergency And Acute Care Electrocardiography. Chichester, West Sussex: Wiley-Blackwell, 2009. Print.
2.) EMCrit, About, Josh Farkas, and the Crew. “Two EKG Patterns Of Pulmonary Embolism Which Mimic MI”. EMCrit. N.p., 2014. Web. 31 Dec. 2015.
3.) Galvagno, Samuel M. Emergency Pathophysiology. Jackson, Wyo.: Teton NewMedia, 2003. Print.
4.) Hqmeded-ecg.blogspot.com,. “Dr. Smith’s ECG Blog: Sudden Severe SOB And ST Segment Elevation: What Is The Diagnosis And Treatment?”. N.p., 2015. Web. 31 Dec. 2015.
5.) Marx, John A et al. Rosen’s Emergency Medicine. Philadelphia: Mosby/Elsevier, 2010. Print.