Are you a healthcare professional currently wondering if your ACLS certification is still valid? Missing your renewal window can lead to stress, missed shifts, or even non-compliance in your workplace. That’s why it’s important to know how long ACLS certification lasts and what you need to do to renew it.
This blog will break down everything you need to know about when ACLS certification expires, including renewal tips to make the process smooth and stress-free.
Image

What Is ACLS Certification?
Imagine this: a patient in cardiac arrest arrives at the emergency room, and the medical team leaps into action with precision and confidence. Behind this quick and effective response is training in Advanced Cardiovascular Life Support (ACLS). For healthcare professionals, ACLS certification is more than just a requirement—it’s a lifesaving tool that empowers them to handle critical emergencies with expertise.
In this blog, we’ll uncover what ACLS certification is, why it’s essential, and how you can achieve this vital credential.
Image

How Long Does BLS Certification Last?
Are you a healthcare professional looking to refresh your Basic Life Support (BLS) certification? Or perhaps you’re a new healthcare worker wondering how long your initial certification will last. Understanding the expiration date of your BLS certification is crucial to maintaining your professional credentials and ensuring you’re prepared to respond to medical emergencies. In this blog post, we’ll delve into the specifics of BLS expiration and renewal so you can stay current with your certification and avoid the hassle of dealing with an expired BLS provider card.
Image
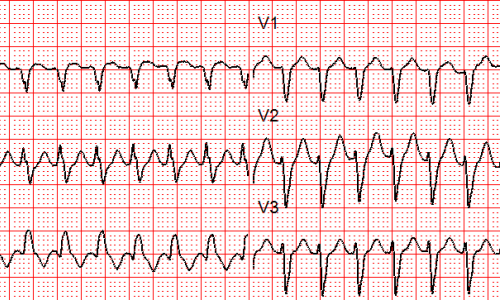
SVT with Aberrancy or Ventricular Tachycardia?
Differentiating between SVT with aberrancy and ventricular tachycardia (VT) can be a challenging task, even for experienced healthcare providers. This blog aims to clarify the distinction between these two cardiac arrhythmias, emphasizing the potential dangers of misdiagnosis and the importance of accurate identification.
Image
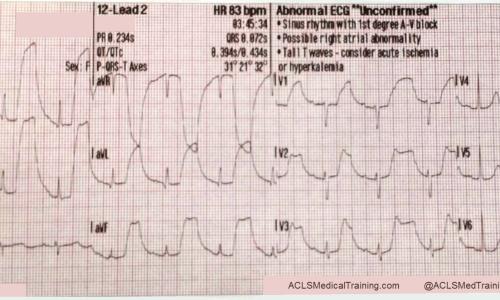
Guide to Understanding ECG Artifact
An electrocardiogram (ECG) is a cornerstone of cardiac diagnosis, providing invaluable insights into the heart’s electrical activity. However, the accuracy of ECG interpretation can be significantly compromised by artifact, which represents any extraneous electrical activity that interferes with the underlying cardiac signal.
